Read More: What is Poisson distribution function? What is the thermal conductivity of air at room temperature? 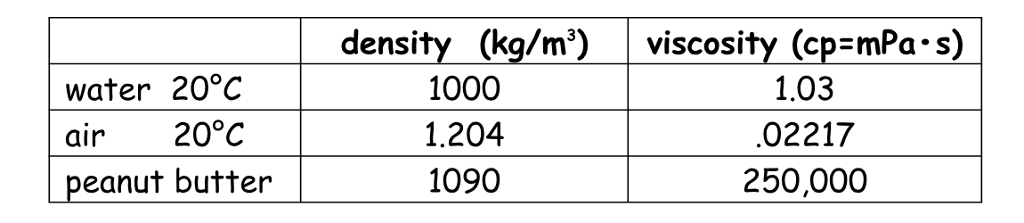
Thermal conductivity (cal/sec)/(cm 2 C/cm) What is the thermal conductivity of still air? Thermal conductivity of Air is 0.025 W/(mK). 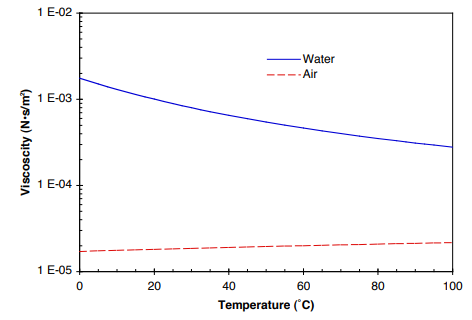
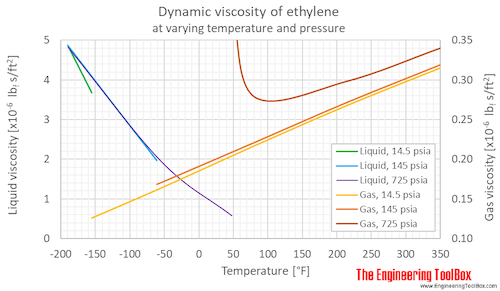
What is the thermal conductivity for air? Water at 20.2 oC (68.4 oF) has the absolute viscosity of one – 1 – centiPoise. How do you calculate dynamic viscosity of air?Įquation (1) is known as the Newtons Law of Friction. There are several formulas and equations to calculate viscosity, the most common of which is Viscosity = (2 x (ball density liquid density) x g x a^2) (9 x v), where g = acceleration due to gravity = 9.8 m/s^2, a = radius of ball bearing, and v = velocity of ball bearing through liquid. The viscosity of a liquid (see Viscosity) is measured using a viscometer, and the best viscometers are those which are able to create and control simple flow fields. What is the standard density of air?ġ.225 kg/m^3 According to the International Standard Atmosphere (ISA) values15 C at sea levelthe density of dry air is at: In Metric units: 1.225 kg/m^3. What is the weight of air at 1 atmosphere of pressure?ġ4.70 pounds per square inch Standard sea-level pressure, by definition, equals 760 mm (29.92 inches) of mercury, 14.70 pounds per square inch, 1,013.25 10 3 dynes per square centimetre, 1,013.25 millibars, one standard atmosphere, or 101.325 kilopascals. The average density of air at NTP is 1.204 kg/m 3 (0.075 pounds per cubic foot). Read More: What vitamin is are only found in foods of animal Charles’ Lawġ.204 kg/m 3 NTP is defined as air at 20 degrees C (293.15 K, 68 degrees F) and 1 atm (101.325 kN/m 2, 101.325 kPa) of pressure. What is its volume at standard pressure? … Boyle’s Law. Example #1: 2.00 L of a gas is at 740.0 mmHg pressure. The one above is just an equation derived from Boyle’s Law. What is the standard pressure in Boyle’s law? … The standard state temperature is 25 degrees C (298 K). STP describes standard conditions and is often used for measuring gas density and volume using the Ideal Gas Law. STP is short for Standard Temperature and Pressure, which is defined to be 273 K (0 degrees Celsius) and 1 atm pressure (or 10 5 Pa). Atmospheric pressure can be measured in atm, torr, mm Hg, psi, Pa, etc. It can be measured using mercury in the equation atmospheric pressure = density of mercury x acceleration due to gravity x height of column of mercury. How do you calculate air pressure?Ītmospheric pressure is the pressure caused by the mass of our gaseous atmosphere. Hence, gaseous viscosity increases with temperature. Since the momentum transfer is caused by free motion of gas molecules between collisions, increasing thermal agitation of the molecules results in a larger viscosity. Online Air Thermal Conductivity Calculator Temperatureĭoes air viscosity change with temperature? What is the thermal conductivity of air at 20 C? = (Pressure * Molecular Weight) / (R * Temp.) … Fugitive Emission Source What is the density of the air for 1 atm and 25 C? What is the pressure of air at 25 degrees Celsius?ġ01.325 kPa SATP – Standard Ambient Temperature and Pressure is a reference with temperature of 25 oC (298.15 K) and pressure of 101.325 kPa. Kinematic viscosity of air at 20C is given to be 1.6 10 – 5 m 2/s.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
